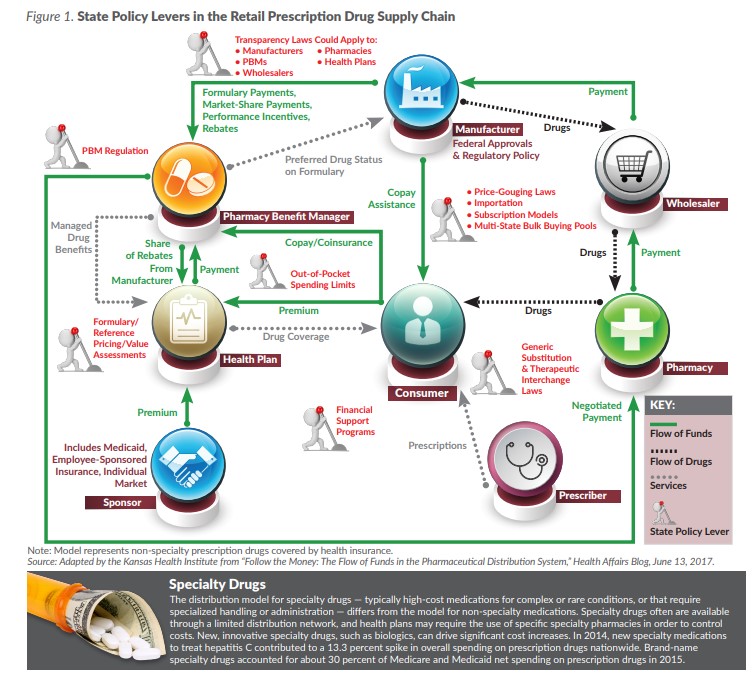
The funding flow is more complex, as it also includes health plans, plan sponsors (i.e., employers and governmental payers) and PBMs. Financial transactions crisscross the ecosystem, sometimes flowing in multiple directions, and often without transparency to consumers or sponsors. That complexity presents a challenge to policymakers who want to design interventions to reduce costs.
Some of the key transactions that state policy can affect include:
-
- Wholesale purchasing: Wholesalers obtain drugs from manufacturers and distribute them to pharmacies. The price paid by wholesalers is sometimes reported as the list price, but it does not account for discounts that they negotiate.
- Formularies: Formularies are lists of brand name and generic medications covered by health plans. Formularies are typically structured into tiers, with drugs given preferred status having lower copays or coinsurance for patients and fewer or no utilization management restrictions, such as prior authorization.
- Reference pricing: As an alternative to formularies, some health plans and plan sponsors set the price they will pay for any drug in a class, with consumers either paying or saving the difference if they select higher- or lower-cost drugs.
- Drug rebates: Manufacturers negotiate rebates with PBMs and health plans to promote use of their drugs, often to achieve preferred drug status on a plan’s formulary. Manufacturers also may negotiate rebates to pharmacies, most commonly large chains. Federally mandated discounts for programs such as Medicaid also may be referred to as rebates.
- PBM pricing: PBMs typically pass most but not all of the value of rebates and discounts through to the health plan or plan sponsor. The portion they retain is sometimes referred to as “spread pricing.” Consumers who are responsible for deductibles or coinsurance calculated on the list price of drugs rather than the price after rebates receive no benefit from the practice. Spread pricing also can refer to the practice of a PBM charging a health plan or plan sponsor more than a pharmacy was paid for a prescription.
- Consumer out-of-pocket costs: Consumers with health coverage pay for prescription drugs through premiums and copays or coinsurance. Some studies have shown that as much as 19 percent of employer-sponsored insurance premiums are attributable to prescription drug costs. Consumers without health coverage, or whose medications are not covered by their health plans, may need to pay the list price out of pocket. Consumers sometimes receive out-of-pocket assistance, such as coupons, from manufacturers.
Federal and State Roles
In the early stages of the supply chain, federal policy holds sway, from approvals of new drugs to protections of intellectual property and requirements for discounts for safety net providers and the joint state-federal Medicaid program. Medicare’s role as the largest purchaser of prescription drugs provides a platform for federal policymakers.
However, states have their own motivations to address the issue. Federal options do not always address state cost and policy concerns, and states have demonstrated they can act as laboratories for innovation.
State Cost Control Strategies
PBMs: In 2018 alone, 21 states passed laws related to PBMs, including bans on contract terms that prevent pharmacies from sharing lower-cost options with consumers and granting explicit permission to disclose out-of-pocket costs with and without insurance coverage. Legislation addressing both of these issues was adopted in Kansas in 2018 (K.S.A. 40-3831). Other states passed laws that included requirements for PBMs to be licensed by the states, restrictions to prevent spread pricing, and requirements that PBMs report pricing and rebate information. In 2020, Kansas House Bill (HB) 2598, which provided for enhanced regulation of PBMs, was amended and passed out of the House Committee on Insurance. However, the substitute bill was stricken from the House calendar and received no further action.
Transparency: A study of state laws passed between 2015-2018 identified 35 laws passed in 22 states (not including Kansas) to increase transparency. Of those, researchers considered only seven passed in six states informative, as they required certain supply chain participants to report profits, net prices or rebates. However, no state had laws that together provided a complete picture of prices and profits across the supply chain, and most focused on only one segment (e.g., manufacturers, PBMs or health plans).
Generic substitution and therapeutic interchange: Nearly all states have laws allowing or mandating pharmacists to substitute lower-cost generic medications for prescribed brand-name medications. Kansas allows pharmacists to dispense a different drug product of the same dosage form and strength and of the same generic names as the prescribed product and requires pharmacists to inform patients of substitution. Kansas also allows pharmacists to substitute an interchangeable biological product (biosimilar), with notice to both the consumer and prescriber.
Some states — including Arkansas, Idaho and Kentucky — go further by allowing therapeutic interchange in community pharmacy settings. Therapeutic interchange differs from generic substitution, as it does not occur between products with the same active ingredients, but instead between products likely to have the same treatment effects.
Medicaid formularies: State Medicaid programs are required to cover all drugs in the federally administered Medicaid Drug Rebate Program if they want to receive mandated discounts (all states do). While the states are ensured statutory minimum discounts (including discounts designed to get states the best price available for brand-name drugs), they typically also negotiate supplemental rebates tied to their programs’ preferred drug lists.
If a state Medicaid program were allowed to exclude certain drugs from its formulary, it would be in a stronger position when negotiating pricing. However, an exclusive (or “closed”) formulary would reduce consumer access to certain medications and limit prescriber options. States have requested waivers to close their Medicaid formularies, but so far none have been approved. However, in January 2020, the federal government signaled it might allow closed formularies under the newly announced Healthy Adult Opportunity block grant initiative.
Value-based rebates and subscription models: New York has implemented a cap on Medicaid prescription drug spending that, when exceeded, triggers value assessments and supplemental rebate targets for high-cost drugs. Since 2018, seven states, including Oklahoma and Colorado, have been approved to negotiate supplemental Medicaid rebates that vary based on clinical outcomes. Other states, including Louisiana and Washington, are instituting “subscription models” in which they receive an unlimited supply of specialty drugs for hepatitis C for a fixed annual price.
Bulk buying pools: To increase their purchasing power, states have formed bulk buying pools for Medicaid, including 29 that are part of the National Medicaid Pooling Initiative, the Top Dollar Program, or the Sovereign States Drug Consortium. Kansas is not a member of a multi-state pool.
Out-of-pocket spending limits: At least 20 states have adopted legislation to limit consumer out-of-pocket expenditures on prescription drugs. While Kansas does not currently have laws limiting cost-sharing on prescription drugs, HB 2557 was introduced in 2020 to limit out-of-pocket costs for insulin to $100 a month. The bill did not get out of committee. Limiting consumer costs on all medications — such as in California’s consumer protection law — or on specific, high-cost medications helps consumers afford prescription drugs they need to manage chronic conditions. Critics of such policies argue that they do not reduce overall costs and instead pass them through to other consumers in the form of higher premiums.
Price gouging: Reports of sudden price increases of critical prescription drugs have motivated some states to propose policies to limit price gouging. The first (and so far only) state law aimed at prohibiting “unconscionable” price increases was adopted in Maryland in 2017. The law was invalidated by the U.S. Fourth Circuit Court of Appeals on constitutional grounds, and the U.S. Supreme Court declined to hear the state’s appeal. In response, Maryland in 2019 enacted a Prescription Drug Affordability Review Board, which can review the cost of a prescription drug and impose limits on payer reimbursements.
Importation: Drug acquisition costs are higher in the United States than in most other countries, including neighboring Canada. Florida, Colorado, Maine and Vermont were the first to propose wholesale importation programs for certain high-cost drugs, and other states have followed suit. HB 2658 was introduced in the 2020 Kansas legislative session to create a prescription drug importation program, but it did not get out of committee. The U.S. Food and Drug Administration recently proposed amending its regulations to allow states to import certain prescription drugs from Canada, and states are awaiting issuance of final regulations to better understand their options.
Conclusion
A challenge for state policymakers who want to address the cost of prescription drugs is developing consensus goals, including determining whether their ultimate intention is to lower consumer costs, state costs, or even costs across the health care system. Some policy options that might achieve one of those goals might have the opposite effect on others. For example, financial support programs for out-of-pocket costs help individual consumers but can increase costs in the aggregate by encouraging use of higher-cost medications.
By developing a consensus goal, policymakers can acknowledge tradeoffs that are implicit in policy options while continuing to build on the lessons learned across the states.